Key Points
- Galvanic corrosion occurs when two metals of differing nobility share an electrolytic environment, causing the less noble metal to corrode.
- The closer two metals are on the galvanic series, the lower the risk of corrosion between them.
- High-risk metal pairings include stainless steel with aluminum, copper with steel, and copper with aluminum.
- Corrosion can be prevented through methods such as dielectric insulation, sacrificial anodes, and zinc coatings.
- Keeping the cathode surface area smaller than the anode surface area helps slow the rate of galvanic reaction.
Galvanic corrosion, also known as dissimilar metal corrosion, is a phenomenon commonly seen in the oil and marine industries as well as in domestic scenarios dealing with damp environments. Galvanic corrosion may occur in thousands of combinations of metals, including those used in washing machines, automobiles, plumbing equipment, and structures of all kinds. Because of this, it’s important to understand which metal combinations could create the most risk potential for galvanic corrosion.
Below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal combinations. We also provide other helpful methods for avoiding galvanic corrosion.
A Brief Overview of Galvanic Corrosion
When two metals of different nobility are joined and kept inside an electrolytic environment, the less noble metal starts to corrode. This phenomenon is known as galvanic corrosion.
The following three factors often contribute to a higher risk of galvanic corrosion:
- Dissimilar metals used in conjunction
- The conductive path
- Electrolyte presence
Although galvanic corrosion often occurs between two metals, it can also occur between two similar metals. For instance, if one section of a bolt is submerged in salt water and the other half is open to the air, an electrostatic potential difference may still occur, leading to galvanic corrosion.
Understanding the galvanic series can help machinists and fabricators understand which two metals are more likely to corrode than others.
Understanding the Galvanic Series
Below, we have included a galvanic series chart comparing different metals and their electrochemical voltage ranges in seawater.
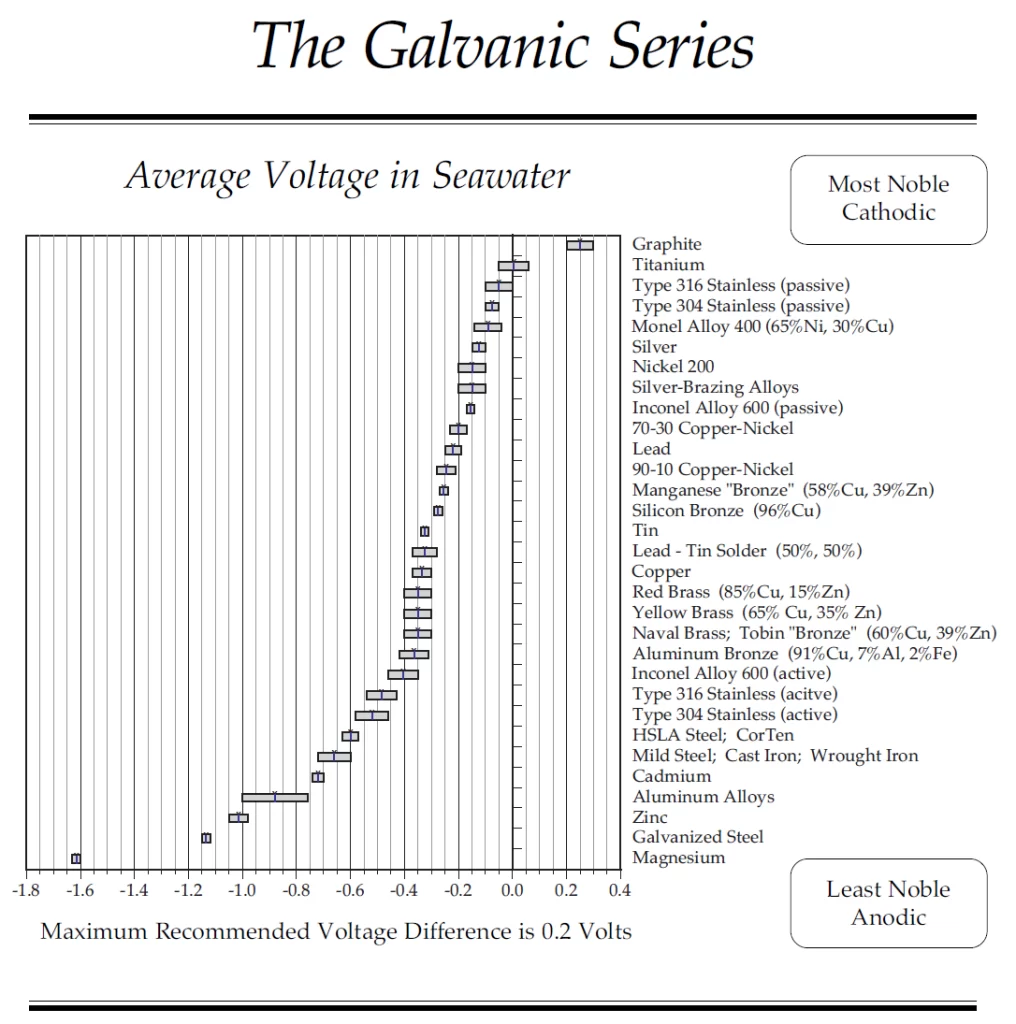
Here, the metal alloys have been arranged from the most noble (at the top) to the least noble (at the bottom); thus, magnesium is the least noble metal, whereas graphite is the highest noble metal in the galvanic series.
For example, imagine two metals are electrically connected to each other under an electrolytic solution. If the current is flowing from metal 1 to metal 2 in the circuit, metal 1 is the cathode and sits atop metal 2 (anode).
Let’s try to simplify it further.
Consider two semi-scratched 316 stainless steel plates (active), one with tin plating and the other with zinc plating, along with their resultant potential as described below.
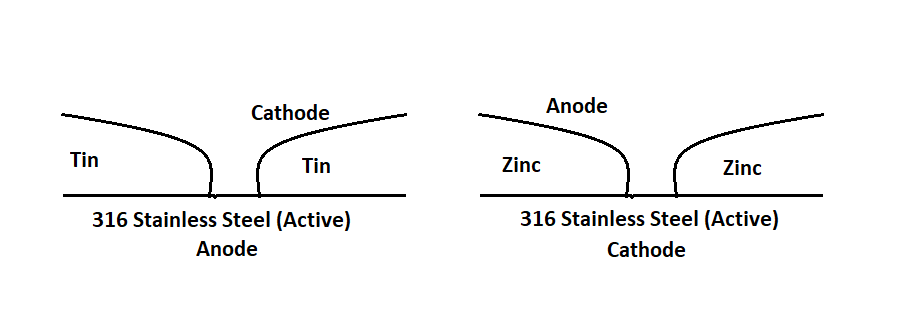
In the first case, due to the high electrostatic potential difference, current flows from tin to stainless steel, i.e., electrons from stainless steel to tin. As the cathode surface area is higher, the corrosion potential of stainless steel is high.
In the second case, zinc lies below stainless steel in the galvanic series, and here, the exposed cathode area is also small. Thus, zinc corrodes in the process but at a much slower rate than in the first case.
To summarize, to avoid galvanic corrosion:
- The choice of different metals must be such that they are close to each other in the galvanic series.
- The area of cathode (high noble metal) must be less than the area of anode (less noble metal).
The effect of corrosion is more at the joint of two dissimilar metals and is less at the remote ends.
|
Buy Certified High-Quality Metals for Industrial Solutions |
||
|
New Aluminum |
New Steel |
New Stainless Steel |
 |
 |
 |
| Reliable supply: Consistent, certified aluminum stock you can trust. High-quality material: Superior-grade aluminum for precision needs. Custom cuts: Accurate cuts down to thousandths of an inch. |
Durability and strength: Reliable for projects needing long-lasting, tough materials. Cost-effective: Quality steel that offers strength without breaking the bank. Custom cutting: Precise cuts tailored to exact needs with top-tier cutting tools. |
Sustainability: Highly eco-friendly as the most recyclable metal on earth. High durability: Resistant to corrosion and wear, ensuring long-lasting performance. Custom precision cuts: Tailored to your specifications with quick, accurate results. |
Avoid These Metal Combinations According to Our Galvanic Corrosion Chart
To mitigate the risk of galvanic corrosion, there are certain metals that should not be used in conjunction. Below, we have provided a galvanic series corrosion chart showcasing the metal combinations that carry the highest risk.
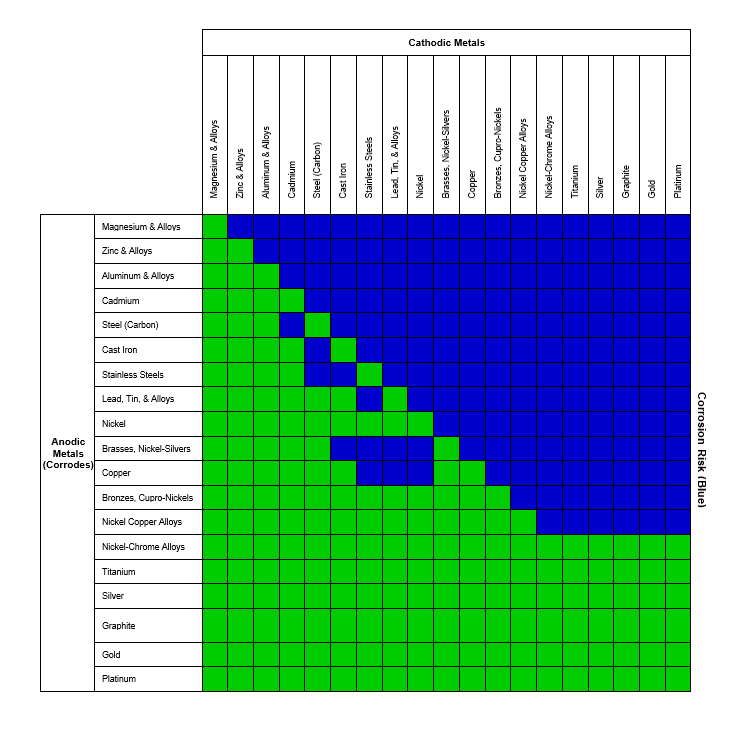 Stainless Steel (Active) + Aluminum
Stainless Steel (Active) + Aluminum
Stainless steel acts as the cathode, and aluminum acts as an anode. Aluminum negatively reacts to stainless steel but with a conducive environment (marine environment); if the surface area of aluminum is larger than stainless steel, corrosion of the aluminum surface is likely to occur. In this regard, there is a risk with hanging heavy aluminum equipment with stainless steel fasteners in any sea vessel. Gradually, the aluminum will start to corrode at the point of coupling and may fall if not quickly amended.
Copper + Steel
Let’s consider a case where a copper water tube is connected to a steel pipe through an adapter, and the water inside is an electrolyte. As steel is electro-negative to copper, electrons will flow from steel to copper, and the steel pipe will corrode. For economic reasons, making an all-copper system can be difficult in most cases. Breaking the electrical contact through a thick six-inch coupling made of insulating material is an easier way to avoid galvanic corrosion.
Copper + Aluminum
As aluminum is lightweight, cheap, and has heat-transfer properties similar to copper, aluminum-copper joints are commonly used in most HVAC applications. Under a non-protective, damp environment, aluminum will act as a sacrificial anode, and copper as a cathode. The aluminum will corrode, leading to joint failures. However, applying zinc coating to the joint saves the aluminum, as the zinc acts as a sacrificial anode to both copper and aluminum.
Tips to Avoid Galvanic Corrosion
While referring to our galvanic corrosion chart is one of the easiest and fastest methods of avoiding dangerous metal combinations, there are other ways of avoiding galvanic corrosion, including the following.
Choose the Right Metals
It’s essential to choose metals with small nobility differences (not more than 0.2 volts). For instance, there is a 0.15-volt difference between nickel and silver, which is fairly acceptable.
Minimize the Cathode Surface Area
The rate of galvanic reaction directly depends on the area of the cathode. Minimizing the large cathode surface area in comparison to the anode will lower the corrosion speed of the cathode.
Coat Cathodes and Anodes With Less Noble Metals
Coating the cathodes and anodes with a less noble metal will protect both from galvanic corrosion. The application of zinc on stainless steels is one of the common examples of galvanic coating.
Use Inhibitors
Nowadays, galvanic corrosion inhibitors are also popular in all industries. It’s a zinc-rich coating that can be applied between the joints of any dissimilar metals and electrical connections. The powerful formula easily absorbs moisture and any energy created by dissimilar metal reactions.
Use Sacrificial Anodes
Using a sacrificial anode that is more electrochemically reactive than the base material helps save the protected material. For instance, instead of zinc coating, a block of zinc positioned close to the galvanized steel plate can save it from galvanic corrosion.
Insulate Dissimilar Metals
Another method of avoiding galvanic corrosion is adding dielectric insulations between the coupling of two dissimilar metals to break the electrical contact. For example, neoprene or nylon washers and bolt sleeves offer full isolation to the stainless steel bolts from aluminum or galvanized steel members. However, when dealing with high-strength connections, you must check the dielectric material against the loading condition.
Purchase Corrosion-Resistant Metals From a Trustworthy Metal Supplier
For more than two decades, Industrial Metal Service has been supplying high-grade alloys to meet your corrosion-resistant design requirements easily. Whether you need new, mill-tested metals or high-quality metal remnants verified with XRF analyzers, we can supply them to your location, anywhere in the country, with no minimum order requirements.
We also offer state-of-the-art metal sawing services to provide you with precision-cut metals for your fabricating and machining needs. Our MetlSaw NF12-T12 can easily cut large, non-ferrous metals up to 12 feet long and 12 inches thick while holding tight tolerances up to ± 0.003″, with vertical and horizontal squareness up to ± 0.0005″/in. thick and ± 0.002″/ft., respectively.
For specialty metals such as titanium, our Amada PCSAW 530 X band saw with pulse cutting technology can cut at a variable speed of 15-120 m/min with a sawing capacity of 30-530 mm for round and square metallic materials.
 Angle
Angle Cast Plate
Cast Plate Diamond Plate
Diamond Plate Flat Bar
Flat Bar Plate
Plate Round Bar
Round Bar Square Bar
Square Bar Square Tubing
Square Tubing Round Tubing
Round Tubing Angle
Angle Channel
Channel Diamond Plate
Diamond Plate I Beam
I Beam Round Bar
Round Bar Sheet
Sheet Square tubing
Square tubing Round Tubing
Round Tubing Rectangular Tubing
Rectangular Tubing Plate
Plate Rectangular Bar
Rectangular Bar Rectangular Tubing
Rectangular Tubing Round Bar
Round Bar Sheet
Sheet Square Bar
Square Bar Square Tubing
Square Tubing
